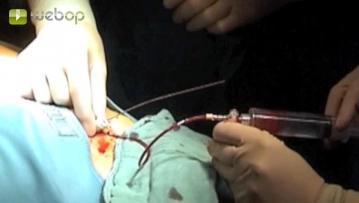
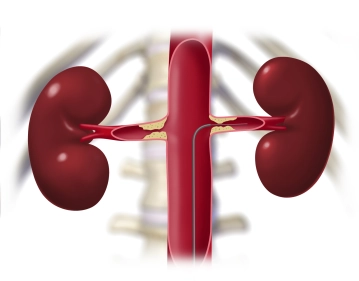
Advance a Terumo® guidewire through the sheath and introduce a pigtail catheter into the suprarenal aorta. Perform survey angiography of the aorta and renal artery in road-mapping technique. Angiography reveals bilateral subtotal stenosis of the renal arteries close to their origins. Next, in this mask place a Renal Double Curve (RDC) catheter into the suprarenal aorta. Slowly retract the guidewire until it slips into the infundibulum at the origin of the left renal artery. Once the guidewire is seated securely, exchange the Terumo® guidewire for a soft-tipped thinner, but harder, metal wire, advancing it carefully into the peripheral renal artery.
Tips:
1. Do not advance the metal wire too far. The resistance must be felt, otherwise the kidney will be perforated, resulting in severe parenchymal hemorrhage.
2. Why does the Terumo® catheter have to be replaced with a harder metal catheter in the first place? This is necessary to obtain sufficient thrust for the balloon-expandable stent to be released in the next surgical step. A soft Terumo® guidewire would dislodge and the stent could not be delivered accurately because it would lack central purchase.
Roadmapping („Pathfinder“):
This involves the administration a small bolus of contrast agent to visualize the abdominal aorta as a roadmap. This image is saved as a mask. Subsequent images are then acquired without contrast media and subtracted from the mask. In this way, for example, only the current position of a radiopaque catheter will be displayed. In the resulting subtraction images, the bright catheter will be visible against the dark background of the abdominal aorta, and any background irrelevant to this study is omitted.